Aluminium is one of the most versatile metals in manufacturing, prized for its lightweight nature, corrosion resistance, and excellent conductivity. A key property that underpins its widespread use is its melting point, which for pure aluminium stands at 660.32°C (1220.58°F). This relatively low melting point compared to metals like steel or copper makes aluminium ideal for processes such as casting, extrusion, and recycling, contributing to its role in industries from aerospace to packaging.
In 2025, global aluminium production reached over 70 million metric tons, with much of it relying on controlled melting for alloying and shaping. This article explores the melting point of aluminium, its variations in alloys, influencing factors, and practical implications.

What is Aluminium Melting Point?
The melting point of a material is the temperature at which it transitions from a solid to a liquid state under standard atmospheric pressure. For pure aluminium, this occurs at precisely 660.32°C (1220.58°F or 933.47 K). This value is crucial because it determines how aluminium behaves in thermal processes. Unlike some metals that have a broad melting range, pure aluminium has a sharp melting point due to its uniform atomic structure—a face-centered cubic (FCC) lattice that requires a specific energy input to disrupt.
In practice, however, aluminium is rarely used in its pure form. Alloying elements introduce variations, often creating a melting range rather than a single point. For instance, the aluminium melting point in Celsius for commercial grades can dip below 600°C, enabling easier processing. This property has historical significance; early attempts to isolate aluminium failed due to its high affinity for oxygen, forming stable compounds like aluminium oxide with melting points exceeding 2000°C.
What Are the Components Affecting Aluminium Melting Point?
Aluminium’s melting point is influenced by its composition and external factors. Pure aluminium consists of 99.99% Al atoms, but impurities or alloying elements alter the atomic bonding, leading to melting point depression—a phenomenon where the addition of foreign atoms disrupts the crystal lattice, reducing the energy needed to melt the material.
Key components include:
- Alloying Elements: Common additions like copper (Cu), magnesium (Mg), silicon (Si), and zinc (Zn) lower the melting point. For example, silicon in casting alloys like A356 reduces it to 555–615°C, improving fluidity for intricate molds.
- Impurities: Trace elements such as iron or oxides can raise or broaden the melting range by creating barriers in atomic movement. High-purity aluminium (99.999%) melts at exactly 660.32°C, while 99.5% pure versions melt around 657°C.
- Pressure and Environment: Under vacuum, the melting point slightly increases due to reduced vapor pressure, while high pressure can elevate it marginally. In manufacturing, atmospheric conditions are controlled to avoid oxidation, which forms high-melting-point aluminium oxide layers.
External factors like heating rate also play a role; rapid heating can cause superheating, where the metal remains solid above its nominal melting point.
What Are the Different Types of Aluminium Alloys and Their Melting Points?
Aluminium alloys are classified into series based on primary alloying elements, each with distinct melting ranges tailored for specific applications. Unlike pure aluminium’s fixed point, alloys exhibit a solidus (start of melting) and liquidus (fully molten) temperature.
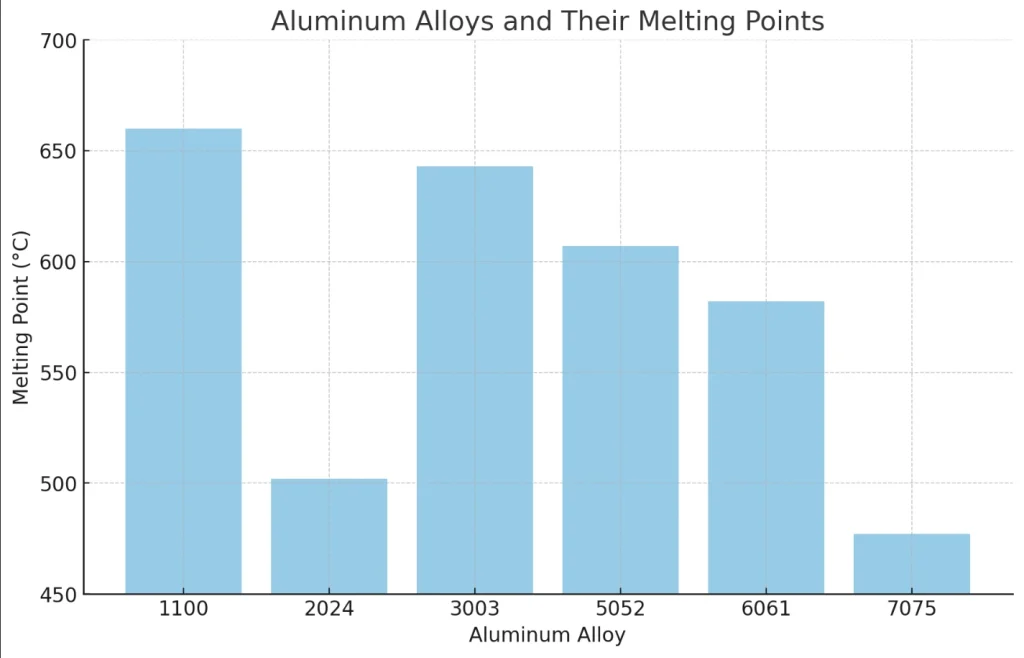
- 1000 Series (Pure Aluminium): Nearly 99% Al, with minimal alloying. Melting point: 643–657°C for alloys like 1100. Used in electrical conductors for high purity and conductivity.
- 2000 Series (Al-Cu Alloys): Copper enhances strength but lowers melting to 500–635°C (e.g., 2024 alloy). Ideal for aerospace structural parts due to high fatigue resistance.
- 3000 Series (Al-Mn Alloys): Manganese improves corrosion resistance. Melting point: 640–655°C (e.g., 3003). Common in beverage cans and heat exchangers.
- 4000 Series (Al-Si Alloys): Silicon boosts castability. Melting range: 550–650°C (e.g., 4043 at 574–632°C). Preferred for welding rods and automotive pistons.
- 5000 Series (Al-Mg Alloys): Magnesium adds weldability. Melting: 570–650°C (e.g., 5052 at 605–650°C). Used in marine applications for saltwater resistance.
- 6000 Series (Al-Mg-Si Alloys): Balanced strength and formability. Melting: 580–650°C (e.g., 6061 at 582–652°C, 6063 at 600–655°C). Widely used in extrusions like window frames.
- 7000 Series (Al-Zn Alloys): Zinc for ultra-high strength. Lowest melting: 475–635°C (e.g., 7075 at 477–635°C). Critical in aircraft wings and sporting goods.
These ranges allow manufacturers to select alloys based on process needs, such as low-melting variants for brazing rods.
What Is the Melting Point of Aluminum? Complete Guide to Temperature & Alloys
What Are the Properties of Aluminium Related to Melting Point?
Aluminium’s melting point intersects with its physical and mechanical properties, influencing manufacturability and performance.
Table 1: Aluminium Properties and Melting Points
| Alloy | Composition (Main Elements) | Density (g/cm³) | Tensile Strength (MPa) | Melting Range (°C) | Applications |
|---|---|---|---|---|---|
| 1100 | 99% Al | 2.71 | 90-110 | 643-657 | Electrical wiring, foil |
| 2024 | Al-Cu (4.5% Cu) | 2.78 | 470 | 500-635 | Aircraft structures |
| 3003 | Al-Mn (1.2% Mn) | 2.73 | 110-190 | 640-655 | Heat exchangers |
| 5052 | Al-Mg (2.5% Mg) | 2.68 | 195-230 | 605-650 | Marine hardware |
| 6061 | Al-Mg-Si (1% Mg, 0.6% Si) | 2.70 | 275-310 | 582-652 | Bicycle frames, automotive parts |
| 7075 | Al-Zn (5.6% Zn) | 2.81 | 570 | 477-635 | Aerospace components |
Data sourced from industry standards. Note that lower melting points often correlate with improved castability but may reduce high-temperature strength.
Aluminium’s thermal conductivity (237 W/m·K for pure) allows efficient heat dissipation, but near melting, it becomes highly reactive, forming oxides that raise effective melting thresholds in impure samples.
History and Production of Aluminium with Melting Point Considerations
Aluminium production has evolved since its discovery in the 19th century, with melting point playing a pivotal role. Ancient civilizations used alum (aluminium compounds) for dyes, but pure aluminium was isolated in 1825 by Hans Christian Ørsted. Its high melting point in oxide form (2072°C) made extraction difficult until the Hall-Héroult process in 1886.
In the Hall-Héroult method, alumina (Al₂O₃) is dissolved in molten cryolite (Na₃AlF₆) to lower the melting point from 2050°C to about 950–980°C, enabling electrolysis. Carbon anodes react to produce aluminium at the cathode. Modern smelters use pre-baked anodes and point feeders for efficiency, considering energy costs tied to maintaining molten baths.
Production today exceeds 70 million tons annually, with China leading at over 50%. Melting considerations include energy efficiency—recycling aluminium requires only 5% of the energy for primary production due to its low melting point.
What Are the Applications of Aluminium Related to Its Melting Point?
Aluminium’s melting point enables diverse applications:
- Casting and Extrusion: Low melting (e.g., 570°C for cast alloys) facilitates die casting for automotive engine blocks and aerospace components.
- Welding and Brazing: Alloys like 4043 (melting 574–632°C) are used in low-temperature joining for ships and vehicles.
- Recycling: Easy re-melting supports sustainable uses in packaging, where aluminium foil (melting 660°C) is recycled efficiently.
- High-Temperature Limits: In electronics, aluminium heat sinks operate below 200°C to avoid softening.
- Aerospace: 7075 alloy’s range (477–635°C) allows forging while maintaining strength for aircraft frames.
These applications leverage the balance between low energy input and durable properties.
What Are the Advantages of Aluminium’s Melting Point?
Aluminium’s relatively low melting point offers several benefits in manufacturing:
- Energy Efficiency: Melting at 660°C requires less energy than steel (1425–1540°C), reducing costs by up to 95% in recycling.
- Processability: Enables casting complex shapes with minimal defects, as in automotive parts.
- Alloy Flexibility: Easy incorporation of elements for tailored properties without extreme temperatures.
- Sustainability: Facilitates closed-loop recycling, minimizing environmental impact.
- Thermal Management: Quick melting and solidification improve production speed in extrusion.
These advantages make aluminium a go-to for lightweight, cost-effective designs.
What Are the Disadvantages of Aluminium’s Melting Point?
Despite its benefits, the melting point presents challenges:
- Limited High-Temperature Use: Softens above 400°C, unsuitable for engines or turbines compared to titanium (1668°C).
- Oxidation Risk: Forms high-melting oxide layers (2072°C), complicating pure melting and requiring fluxes.
- Alloy Variability: Broad melting ranges can lead to inconsistent solidification, causing porosity in casts.
- Energy Sensitivity: Impurities raise effective melting, increasing energy needs and defect risks.
- Strength Trade-Off: Lower melting often correlates with reduced creep resistance at elevated temperatures.
Manufacturers mitigate these with precise controls and alloy selections.
Aluminium oxide
What Is the Maximum Operating Temperature of Aluminium?
Aluminium’s maximum service temperature is typically 200–300°C for most alloys, well below melting to prevent creep or loss of strength. For high-heat applications, creep-resistant alloys like 2618 operate up to 250°C.
Table 2: Maximum Operating Temperatures for Aluminium Alloys
| Alloy | Solidus (°C) | Liquidus (°C) | Max Operating Temp (°C) |
|---|---|---|---|
| 2024 | 500 | 635 | 200 |
| 6061 | 582 | 652 | 180 |
| 7075 | 477 | 635 | 150 |
| A356 | 555 | 615 | 220 |
Data from material databases. Exceeding these leads to annealing and weakening.
How Strong is Aluminium Near Its Melting Point?
As temperature approaches melting, aluminium’s strength plummets due to increased atomic mobility. At room temperature, 7075 alloy reaches 570 MPa tensile strength, but at 400°C, it drops below 100 MPa. Pure aluminium is ductile near melting, aiding forming but risking deformation in service.
Is Aluminium Safe to Use Considering Melting Point?
Aluminium is generally safe, but melting processes require precautions against burns and fumes. Molten aluminium can cause explosive reactions with water, and oxide dust is inhalable. Alloys comply with REACH/RoHS, but leaded variants need handling care. No major toxicity issues, though allergies to dust occur rarely.
Does Aluminium Violate REACH or RoHS?
Most aluminium alloys comply with REACH (Registration, Evaluation, Authorisation of Chemicals) and RoHS (Restriction of Hazardous Substances), as they avoid restricted elements like lead in high concentrations. Exemptions exist for specific uses, but processing hazards like HF from cryolite in production require mitigation.
Aluminium Melting Point vs. Other Metals
Aluminium’s melting point (660°C) is moderate, higher than sodium (98°C) and magnesium (650°C) but lower than silicon (1414°C), steel (1425–1540°C), and copper (1085°C).
- Vs. Sodium: Aluminium’s higher melting reflects stronger metallic bonds from three valence electrons vs. sodium’s one.
- Vs. Magnesium: Slightly higher due to smaller atomic radius and denser packing.
- Vs. Silicon: Silicon’s covalent network requires more energy to break.
- Vs. Steel/Copper: Lower melting aids aluminium’s use in lightweight alternatives, though with less heat resistance.
This positions aluminium for energy-efficient applications where high melting isn’t needed.
Frequently Asked Questions About Aluminium Melting Point
Why does aluminium oxide have a high melting point?
Aluminium oxide (Al₂O₃) melts at 2072°C due to strong ionic bonds between Al³⁺ and O²⁻ ions in its hexagonal close-packed structure. This requires immense energy to disrupt, unlike metallic aluminium.
Why does aluminium sulfide have a high melting point?
Aluminium sulfide (Al₂S₃) has a melting point around 1100°C, stemming from ionic-covalent bonding, though lower than oxide due to larger sulfide ions reducing lattice energy.
Why does magnesium have a higher melting point than aluminium?
Magnesium melts at 650°C, slightly lower than aluminium’s 660°C. Earlier misconceptions arise from alloy comparisons; pure Mg has weaker bonds.
Why does silicon have a higher melting point than aluminium?
Silicon’s 1414°C melting point results from its covalent diamond structure, forming strong directional bonds vs. aluminium’s metallic delocalized electrons.
Why does sodium have a lower melting point than aluminium?
Sodium’s 98°C melting point is due to single valence electron contributing to weaker metallic bonding compared to aluminium’s three.
Why is the melting point of aluminium oxide so high?
The high charge density of Al³⁺ and O²⁻ creates robust electrostatic attractions in the corundum lattice.
Why is the melting point of silicon higher than aluminium?
As above, covalent vs. metallic bonding.
Why melting point of aluminium is higher than sodium?
More valence electrons strengthen the metallic bond.
Why melting point of aluminium oxide lower than magnesium oxide?
Magnesium oxide (2852°C) has higher lattice energy from Mg²⁺’s smaller size and full ionic character, while Al₂O₃ has partial covalent traits.
Why the melting point of sodium is less than aluminium?
Fewer delocalized electrons in sodium.

David Li
David Li is the CNC Machining Expert at Cncpioneer, with 6 years of frontline experience as a CNC programmer, process engineer, and precision machining specialist. He excels in translating complex machining challenges into clear, actionable advice for operators, engineers, and manufacturers.